Men with oligospermia did not experience further decline. 5 Additionally, the increase may be due to the increased abstinence time before the second sample. While these results showed statistically significant increases in all sperm parameters, the magnitude of change is within normal individual variation and may be influenced by regression to the mean. Because the vaccines contain mRNA and not the live virus, it is unlikely that the vaccine would affect sperm parameters. In this study of sperm parameters before and after 2 doses of a COVID-19 mRNA vaccine, there were no significant decreases in any sperm parameter among this small cohort of healthy men. The waterfall plot shows the within-participant change in TMSC from baseline (range, −22 million to 93 million) for each man ( Figure). No man became azoospermic after the vaccine. Of these 8, 7 men had increased sperm concentration to normozoospermic range at follow-up (median concentration, 22 million/mL ), and 1 man remained oligospermic. Semen volume and sperm motility also significantly increased ( Table).Įight of the 45 men were oligospermic before the vaccine (median concentration, 8.5 million/mL ). After the second vaccine dose, the median sperm concentration significantly increased to 30 million/mL (IQR, 21.5-40.5 P = .02) and the median TMSC to 44 million (IQR, 27.5-98 P = .001). Baseline median sperm concentration and TMSC were 26 million/mL (IQR, 19.5-34) and 36 million (IQR, 18-51), respectively. Baseline samples were obtained after a median abstinence period of 2.8 days (IQR, 2-3) and follow-up samples after a median of 3 days (IQR, 3-4). 
A 2-tailed P < .05 was considered statistically significant.īetween December 17, 2020, and January 12, 2021, 45 men volunteered (median age, 28 years ) follow-up samples were obtained at a median of 75 days (IQR, 70-86) after the second dose. Statistical analysis was performed with SPSS version 24 (IBM). The Wilcoxon rank sum test was used to compare pre- and postvaccination semen parameters. After calculating data distribution on normality test, medians and interquartile ranges (IQRs) were reported for all variables. 4 Individuals with oligospermia (sperm concentration <15 million/mL) were included. Semen analyses were performed by trained andrologists per World Health Organization guidelines and included semen volume, sperm concentration, sperm motility, and total motile sperm count (TMSC). Participants provided a semen sample after 2 to 7 days of abstinence, prior to receiving the first vaccine dose and approximately 70 days after the second. 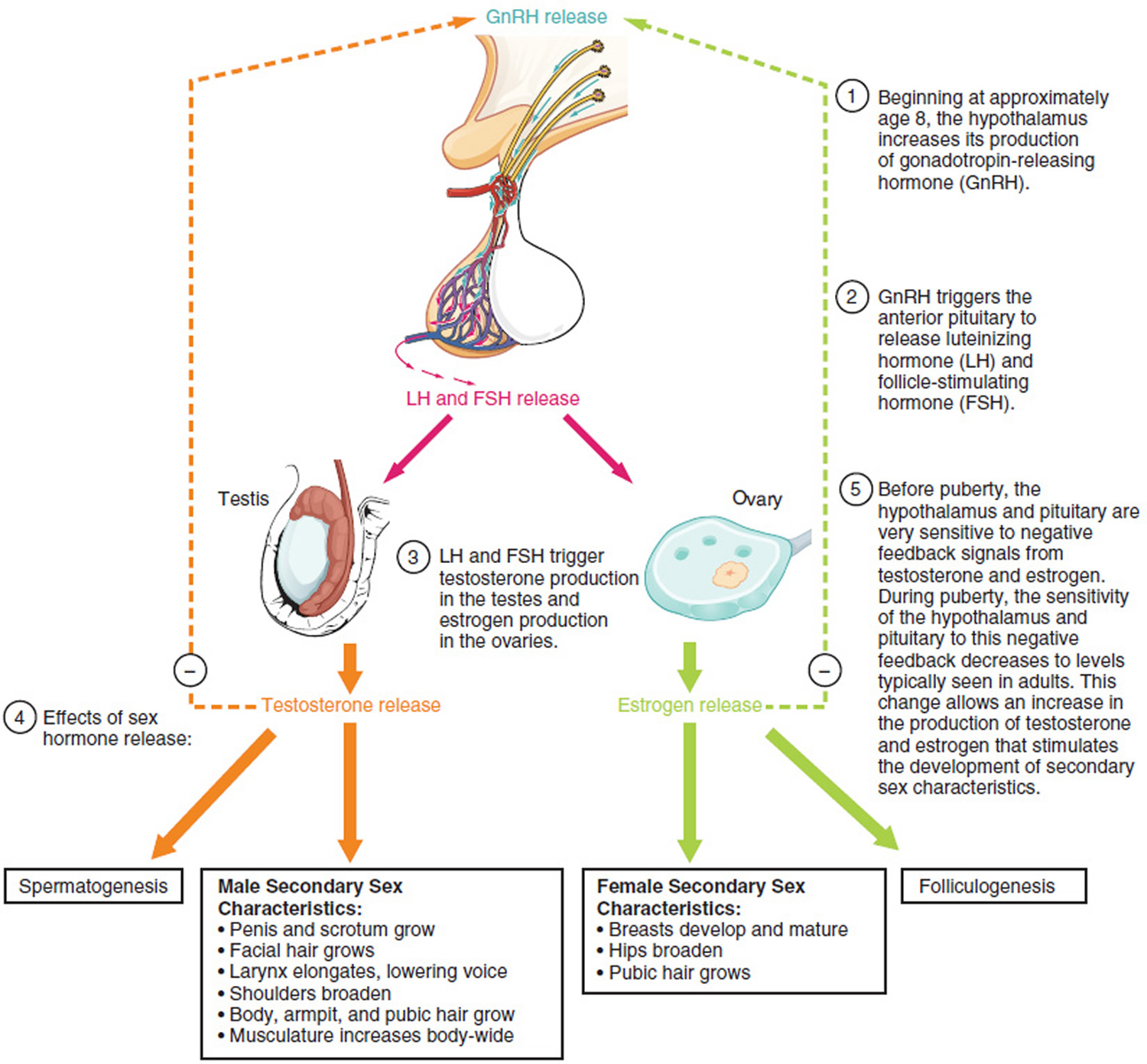
Those with COVID-19 symptoms or a positive test result within 90 days were excluded. Men were prescreened to ensure they had no underlying fertility issues. The University of Miami institutional review board approved the study and written informed consent was obtained from all participants. This single-center prospective study at the University of Miami recruited healthy volunteers aged 18 to 50 years scheduled for mRNA COVID-19 vaccination through flyers posted throughout the university hospital and internal list-serve emails. Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment.Clinical Implications of Basic Neuroscience. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
